Mixtures and Separation
Matthew Williams
||6 min readPaper 1 LikelyCSEC ChemistryMixturesSeparation Techniques
Exam-focused notes on mixtures, solutions, and separation techniques
Overview
Matter can be classified into two main groups: pure substances and mixtures.
A pure substance contains only one type of material and has fixed properties, while a mixture consists of two or more substances physically combined in variable proportions. Understanding this distinction is essential, as it determines how substances behave and how they can be separated.
Pure Substances and Mixtures
A pure substance has a fixed composition and constant properties. It cannot be separated into simpler substances by physical means. Pure substances are divided into elements and compounds.
An element is the simplest form of matter and consists of only one type of atom. It cannot be broken down by ordinary chemical or physical methods. Examples include iron, copper, and oxygen.
A compound consists of two or more different elements chemically combined in fixed proportions. Its properties differ from those of the individual elements. For example, water is composed of hydrogen and oxygen, but its properties are very different from those gases.
A mixture, in contrast, consists of substances that are not chemically combined. Each component retains its own properties and can be separated by physical methods. The composition of a mixture is not fixed.
Homogeneous and Heterogeneous Mixtures
Mixtures can be further classified based on their uniformity.
A homogeneous mixture has a uniform composition throughout, and its components cannot be distinguished. A solution is a common example, such as salt dissolved in water or air.
A heterogeneous mixture has a non-uniform composition, and its components can be distinguished. Examples include sand and salt or muddy water.
Solutions, Suspensions, and Colloids
Different types of mixtures can be distinguished based on particle size and behaviour.
-
Solution: A homogeneous mixture in which a solute is dissolved in a solvent. The particles are very small, do not settle, and cannot be separated by filtration. Solutions are usually transparent.
-
Suspension: A heterogeneous mixture containing visible particles. These particles settle over time and can be separated by filtration.
-
Colloid: A heterogeneous mixture with particles intermediate in size between those of a solution and a suspension. The particles do not settle but can scatter light.
These differences arise from particle size and how particles interact with the surrounding medium.
Solubility
Solubility is the mass of solute that can dissolve in a fixed amount of solvent at a given temperature, usually expressed as grams per 100 g of water.
A solution becomes saturated when it contains the maximum amount of solute that can dissolve at a given temperature. Any additional solute remains undissolved.
For most solid solutes in water, solubility increases as temperature increases. When a saturated solution is cooled, excess solute may crystallise out.
Types of Solutions
Solutions are not limited to a solid dissolved in a liquid. Any two substances can potentially form a solution, depending on their compatibility:
| Type | Example |
|---|---|
| Solid in liquid | Salt dissolved in water |
| Liquid in liquid | Ethanol dissolved in water |
| Gas in liquid | Carbon dioxide dissolved in water (fizzy drinks) |
| Solid in solid | Brass (copper and zinc — a solid alloy) |
| Gas in gas | Air (nitrogen, oxygen, and other gases) |
The most common solutions encountered in chemistry are solids or gases dissolved in a liquid solvent.
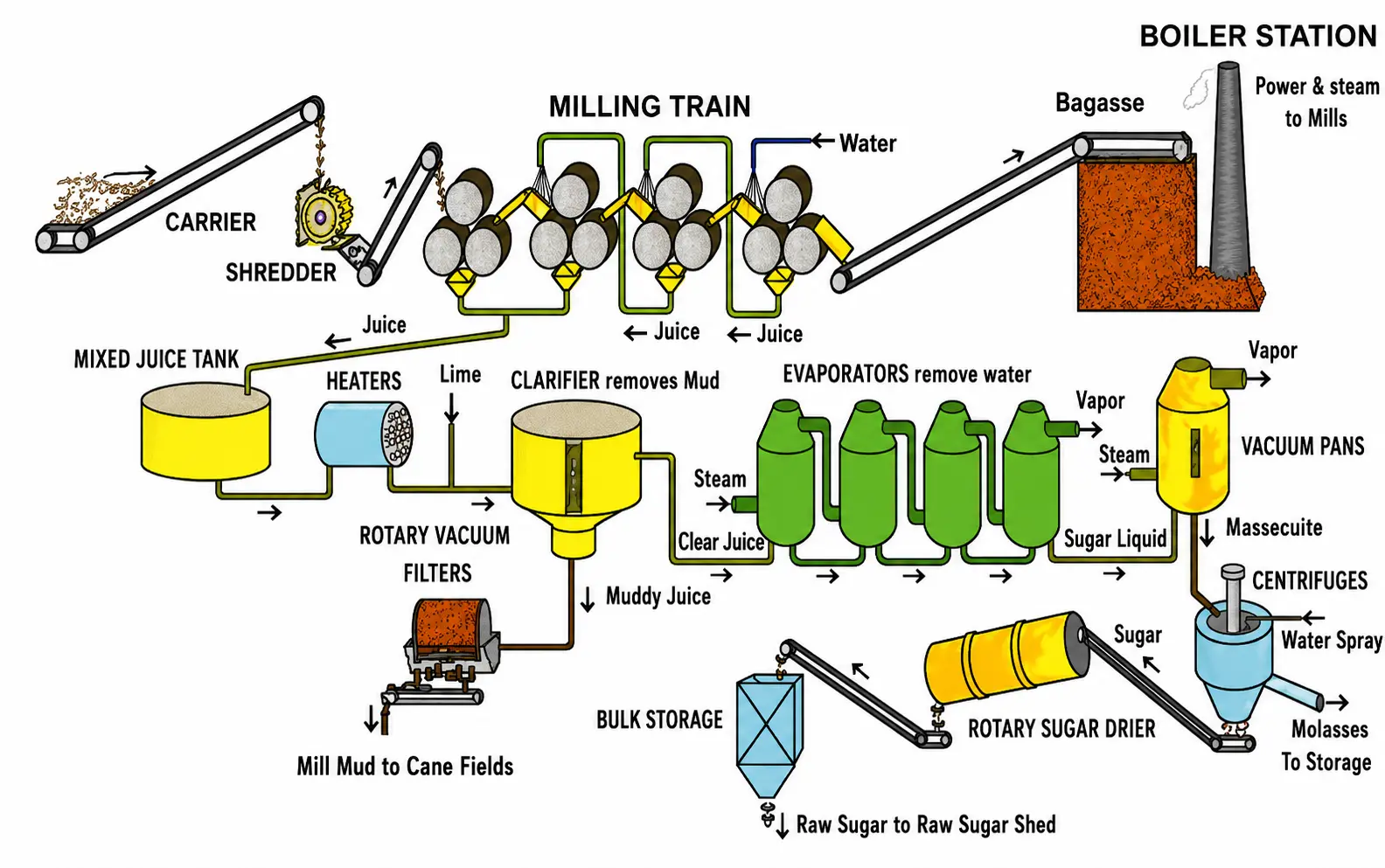
Industrial Extraction of Sucrose from Sugar Cane
The extraction of sucrose from sugar cane is a Caribbean industrial process that puts all of these separation techniques to work in sequence.
The extraction of sucrose (table sugar) from sugar cane is an important Caribbean industrial process that applies several separation techniques in sequence.
-
Crushing — the harvested cane stalks are crushed between heavy rollers to squeeze out the raw juice, which contains sucrose along with impurities such as mud, fibres, and colouring matter.
-
Precipitation and filtration — milk of lime (calcium hydroxide solution) is added to the juice. It reacts with impurities, causing them to precipitate. The mixture is filtered to remove these solid impurities, leaving a clearer juice.
-
Vacuum distillation (evaporation) — the clarified juice is heated under reduced pressure (vacuum) so that water evaporates at a lower temperature. This concentrates the sucrose solution without decomposing the sugar.
-
Crystallisation — as the concentrated syrup cools, sucrose crystallises out of solution. The crystals grow larger as more of the solution solidifies.
-
Centrifugation — the mixture of crystals and remaining syrup (molasses) is spun at high speed in a centrifuge. The force separates the solid sugar crystals from the liquid molasses.
The result is raw sugar, which may be further purified (refined) to produce the white table sugar used in food.
Exam Tip
For the sugar extraction question, know the stages in order: crushing → precipitation/filtration → vacuum distillation → crystallisation → centrifugation. The syllabus may also ask you to identify which separation technique is being applied at each stage.
Separating Mixtures
Mixtures can be separated because their components have different physical properties such as particle size, boiling point, density, and solubility.
-
Filtration: Used to separate an insoluble solid from a liquid. The solid remains as the residue, while the liquid passes through as the filtrate.
-
Evaporation: Used to obtain a dissolved solid by heating the solution so that the solvent evaporates.
-
Crystallisation: Used to obtain a solid in crystalline form by allowing the solvent to evaporate slowly.
-
Simple distillation: Used to separate a liquid from a solution. The liquid is vaporised and then condensed to form the distillate.
-
Fractional distillation: Used to separate two or more miscible liquids with different boiling points.
-
Separating funnel: Used to separate immiscible liquids based on differences in density.
-
Chromatography: Used to separate dissolved substances based on differences in solubility and attraction to a medium.
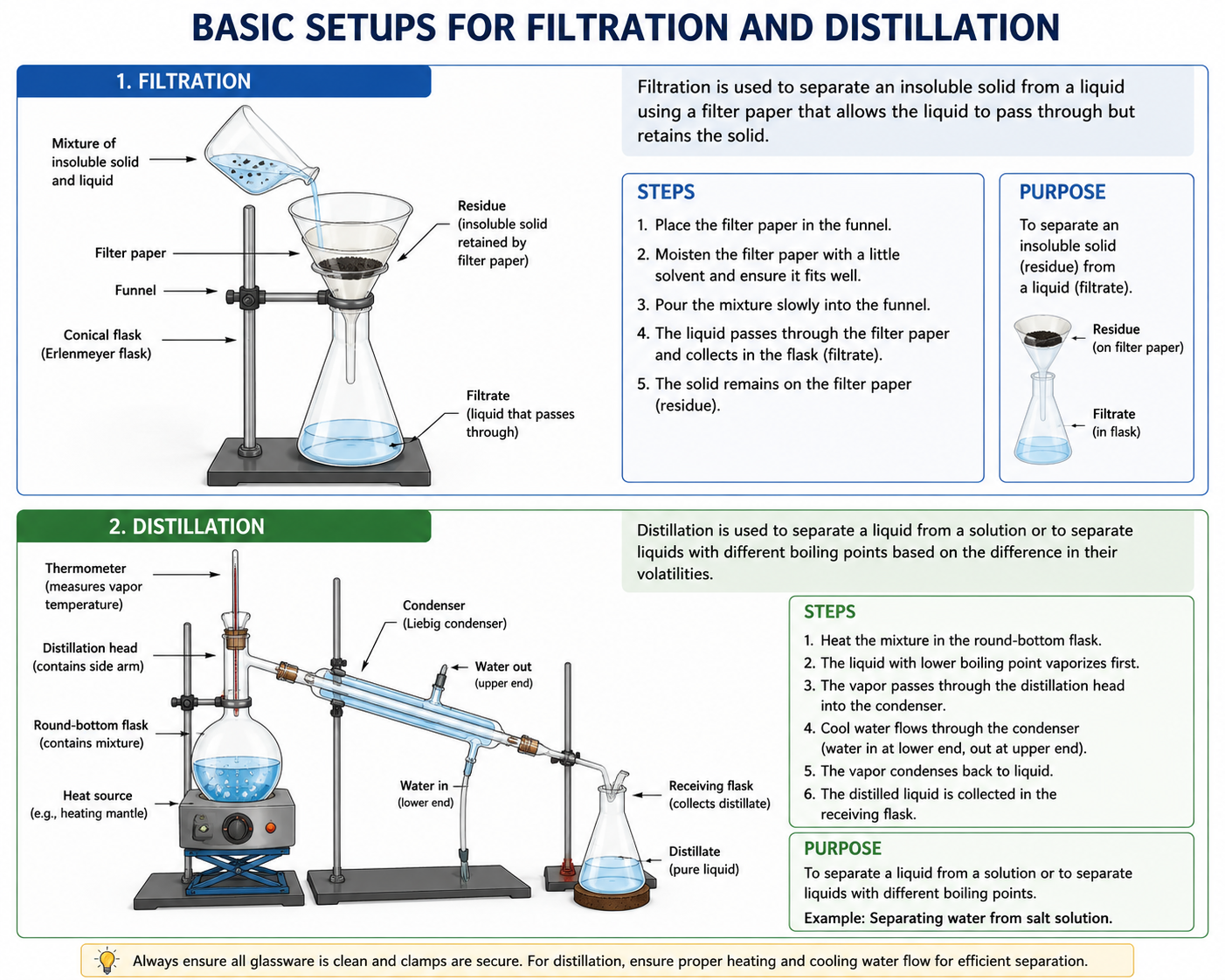
Each method depends on a specific physical property, so choosing the correct method requires identifying how the components differ.
Summary of Key Principles
- Matter is classified into pure substances and mixtures
- Elements and compounds are pure substances with fixed composition
- Mixtures have variable composition and can be separated physically
- Homogeneous mixtures are uniform; heterogeneous mixtures are not
- Solutions, suspensions, and colloids differ by particle size and behaviour
- Separation methods depend on differences in physical properties
A clear understanding of these distinctions allows for correct identification and separation of substances in both theory and practical applications.