States of Matter
Matthew Williams
||4 min readCSEC ChemistryParticulate TheoryStates of Matter
Exam-focused notes on the particulate nature of matter, states of matter, and changes of state
Overview
Matter is anything that has mass and occupies space.
All matter exists in three main states: solid, liquid, and gas. The differences between these states are explained by the particulate theory of matter, which describes how particles are arranged, how they move, and how they interact.
The Particulate Theory of Matter
The particulate theory states that:
- all matter is made of particles
- particles are in constant random motion
- there are spaces between particles
- there are forces of attraction between particles
This theory explains the physical behaviour of substances, including their properties and changes of state.
Temperature directly affects particle motion. As temperature increases, particles gain kinetic energy and move faster. When temperature decreases, particles lose energy and move more slowly.
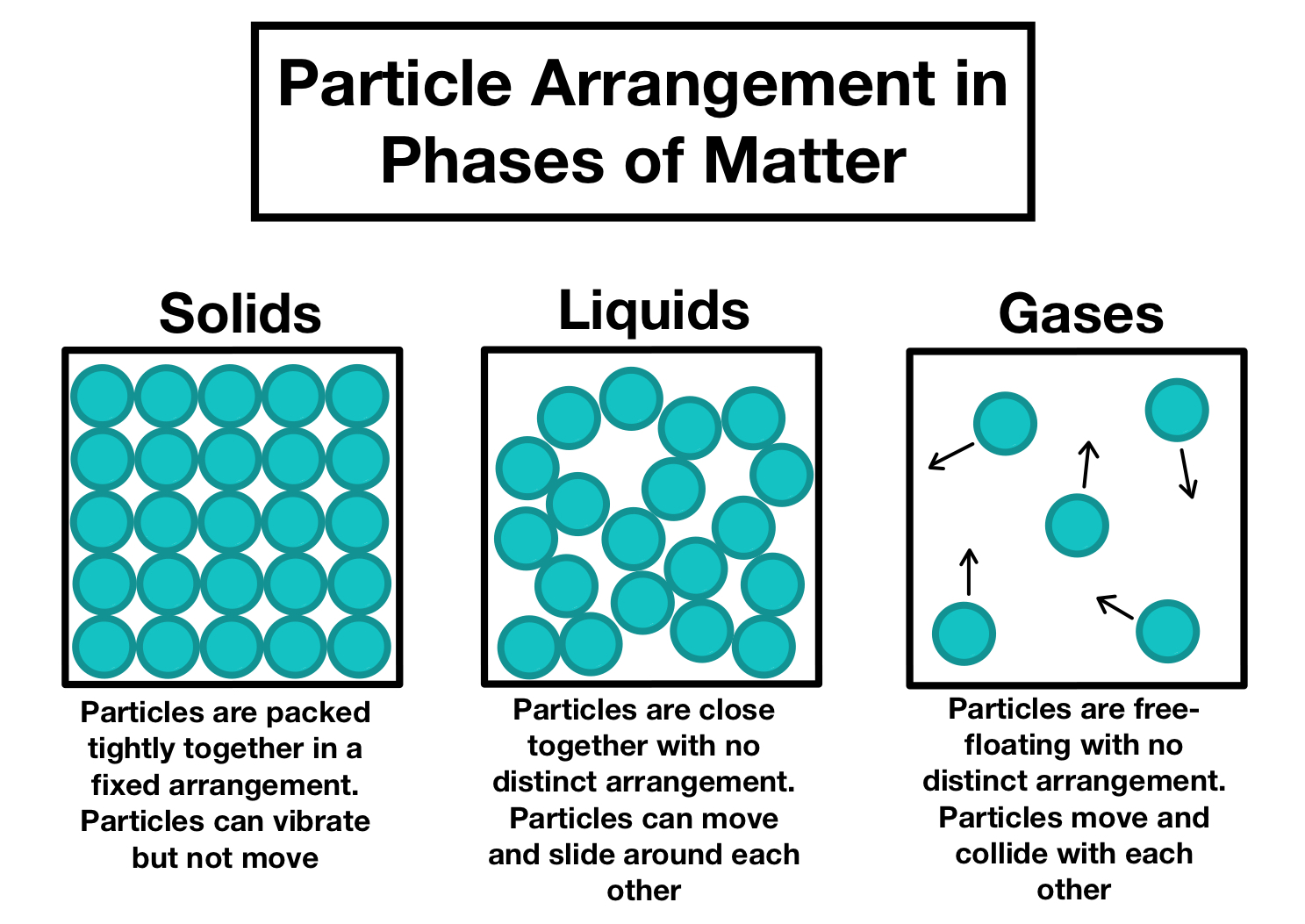
The Three States of Matter
The properties of solids, liquids, and gases can be explained in terms of particle arrangement, energy, and forces of attraction.
In a solid, particles are packed closely together in fixed positions and can only vibrate. Strong forces of attraction hold them in place, giving solids a fixed shape and volume and making them difficult to compress.
In a liquid, particles are still close together but are not fixed. They have enough energy to move past each other, which allows liquids to flow and take the shape of their container while maintaining a fixed volume. The forces of attraction are weaker than in solids.
In a gas, particles are far apart and move rapidly in all directions. The forces of attraction are very weak, so gases have no fixed shape or volume and expand to fill any container. They are easily compressed because of the large spaces between particles.
Changes of State
A change of state occurs when a substance changes from one state to another due to a change in temperature. This is a physical change, meaning the substance itself does not change.
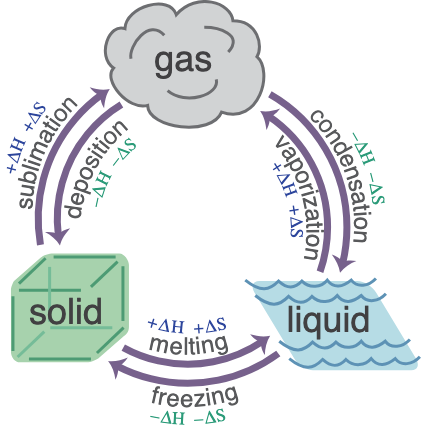
-
Melting (solid → liquid): When a solid is heated, particles gain energy and overcome the forces holding them in fixed positions, allowing them to move more freely.
-
Freezing (liquid → solid): When a liquid is cooled, particles lose energy and become fixed in place as forces of attraction strengthen.
-
Evaporation (liquid → gas): Particles at the surface gain enough energy to escape into the gas state. This can occur at any temperature.
-
Boiling (liquid → gas): At the boiling point, particles throughout the liquid gain enough energy to form a gas. The temperature remains constant during this process.
-
Condensation (gas → liquid): When a gas is cooled, particles lose energy and move closer together to form a liquid.
-
Sublimation (solid ↔ gas): Some substances change directly between solid and gas without passing through the liquid state.
Heating Behaviour
When a substance is heated, its temperature increases as particles gain kinetic energy. However, during melting and boiling, the temperature remains constant for a period of time.
Hover a segment to learn more.
This occurs because the energy supplied is used to overcome the forces of attraction between particles rather than increase their speed.
A cooling curve shows the reverse process. When a substance cools, its temperature drops steadily until it reaches the boiling point, where it stays constant while the gas condenses to a liquid. The temperature then drops again until it reaches the melting point, where it stays constant again while the liquid solidifies. These flat sections (plateaus) appear at the same temperatures as on the heating curve — boiling point and melting point are the same regardless of whether the substance is heating or cooling.
Evidence for the Particle Model
The particulate nature of matter is supported by observable processes such as diffusion and osmosis.
-
Diffusion: The movement of particles from a region of higher concentration to a region of lower concentration. This shows that particles are in constant motion and that spaces exist between them.
-
Osmosis: The movement of water molecules through a partially permeable membrane from a dilute solution to a more concentrated solution. This is a special case of diffusion.
Summary of Key Principles
- Matter is made of particles
- Particle arrangement and energy determine the state of matter
- Temperature affects particle motion
- Changes of state occur when energy is added or removed
- Physical properties can be explained using the particle model
A clear understanding of particle behaviour allows for accurate explanation of most questions on this topic.